 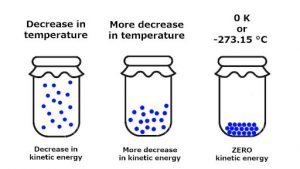
We can see in subfigure 2.Entropy is a scientific concept, as well as a measurable physical property, that is most commonly associated with a state of disorder, randomness, or uncertainty. For example, in figure 1, the atoms involved in the reaction are C, H, and O, each of which are represented in the intermediate state in elemental form. 
This is a useful intermediate state since it can be used for any possible chemical reaction. In subfigure 2.2, we consider one such possible path, consisting of two reactions passing through an intermediate state containing all the atoms involved in the reaction, each in elemental form. Since H is a state function, we can follow any path from R to P and calculate ΔH along that path. 
We wish to calculate the heat absorbed or released in this reaction, which is ΔH. Consider the prototypical reaction in subfigure 2.1, with reactants R being converted to products P. Now, the existence of an energy state function H is of considerable importance in calculating heats of reaction. Since the elevation thus a state function, the elevation gain is independent of the path. Our elevation, standing on the third floor, is independent of how we got to the third floor, and the same is true of the first floor. This is simply because the elevation is a "state function". Each path produces exactly the same elevation gain, even though the distance traveled is significantly different from one path to the next. We could even walk outside and have a crane lift us to the roof of the building, from which we climb down to the third floor. We can simply climb up two flights of stairs, or we can climb one flight of stairs, walk the length of the building, then walk a second flight of stairs. This difference is independent of the path we choose to get from the first floor to the third floor. Consider the difference in elevation between the first floor and the third floor of a building. The concept of a state function is somewhat analogous to the idea of elevation. 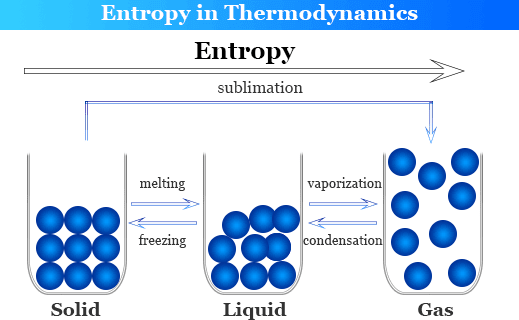
H, which we call the enthalpy, is a state function, since its value depends only on the state of the materials under consideration, that is, the temperature, pressure and composition of these materials. We choose this function, H, so that the change in the function, ΔH = H products - H reactants, is equal to the heat of reaction q under constant pressure conditions. Likewise, the value of this energy function in the product state is independent of how the products are prepared. Were this not the case, we could endlessly produce unlimited quantities of energy by following the circuitous path which continually reproduces the initial reactants.īy this reasoning, we can define an energy function whose value for the reactants is independent of how the reactant state was prepared. Therefore, we cannot extract any energy from the reactants by a process which simply recreates the reactants. This is a statement of the conservation of energy: the energy in the reactant state does not depend upon the processes which produced that state. We discover that the net heat transferred (again provided that all reactions occur under constant pressure) is exactly zero. A consequence of our observation of Hess's Law is therefore that the net heat evolved or absorbed during a reaction is independent of the path connecting the reactant to product (this statement is again subject to our restriction that all reactions in the alternative path must occur under constant pressure conditions).Ī slightly different view of figure 1 results from beginning at the reactant box and following a complete circuit through the other boxes leading back to the reactant box, summing the net heats of reaction as we go. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
